Change Management
Change Management is the process of introducing technical or organisational change within a company in a controlled manner.
QbD’s business experts help you to introduce and efficiently mange change within your organisation.

Change control
Document management is of vital importance especially in highly regulated environments. In the pharmaceutical and medical device sectors it is extremely important to exercise strict control over documents and processes. To this end companies must ensure that they have adequate change control systems in place. In today’s age of electronic media change control has been greatly assisted by the introduction of software solutions. Numerous commercial software solutions exist. However, companies must be able to make the right decision about which software is most appropriate for them.
Organisational changes
There are several methods and models in use for change implementation including, the approach to achieve this may be directive or participative.
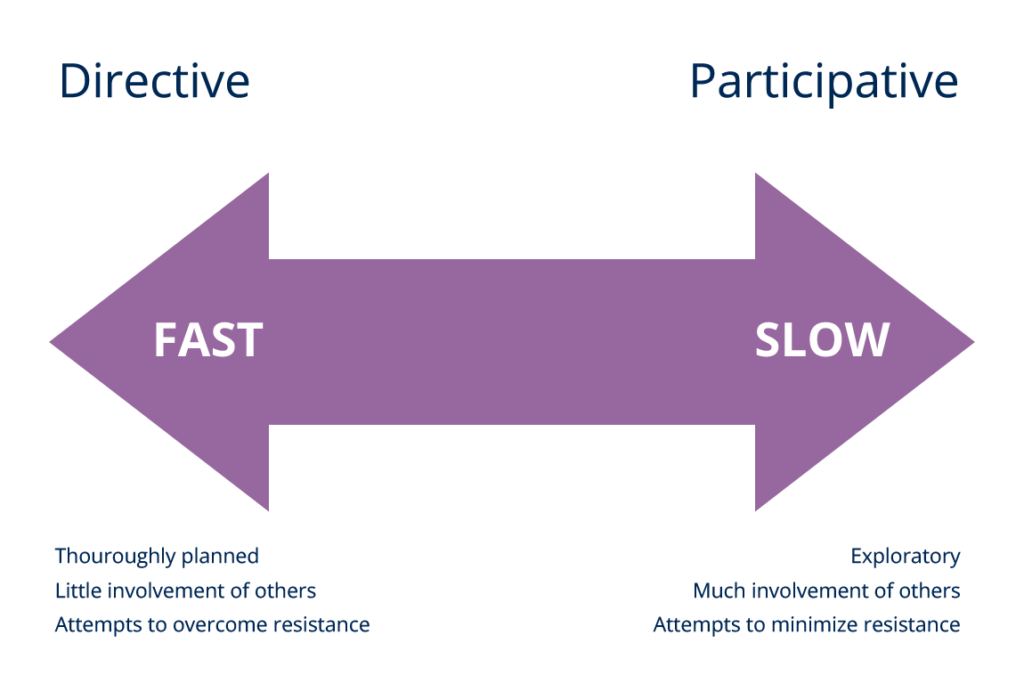
- Directive strategies to change “impose” change through formal channels of management authority with little or no involvement of other people.
- Participative strategies on the other hand, although usually initiated by management, is based on involving people in order to win their commitment and ability to influence the change process by giving them more autonomy.
- Unfreeze-Change-Freeze
- PCI (People Centered Implementation)
- ADKAR (developed by Prosci)
- Awareness – of why the change is needed
- Desire – to support and participate in the change
- Knowledge – of how to change
- Ability – to implement new skills and behaviors
- Reinforcement – to sustain the change
How we support you
- Choosing the right change model for your company
- Implementing a successful change management process
- Advice on available software solutions on offer
- Making an informed choice of which software may be most appropriate for your company
- Leading the software installation and validation process including IQ, OQ and PQ
- Writing appropriate SOPs
- Training your staff and creating enthusiasm and participation to further the change implementation process
- Setting up a change control system also in the absence of appropriate software
Why opt for the QbD Group?
⟶ Expert Advice from our team of business consultants
10+ years of experience
Benefit from our team of highly qualified and experienced business experts.
Global presence
QbD operates globally, at QbD Austria with a special focus on Europe (EU) and China.
Eye level communication
Clear and transparent objectives, and constant evaluation.
Contact us
Talk to an expert
Contact us for more information or to request a free, no-obligation proposal.
